Airports Frankfurt and Bangalore establish partnership
Feb 27, 2026 at 12:59 PM
Renault Trucks maintains market share 2025
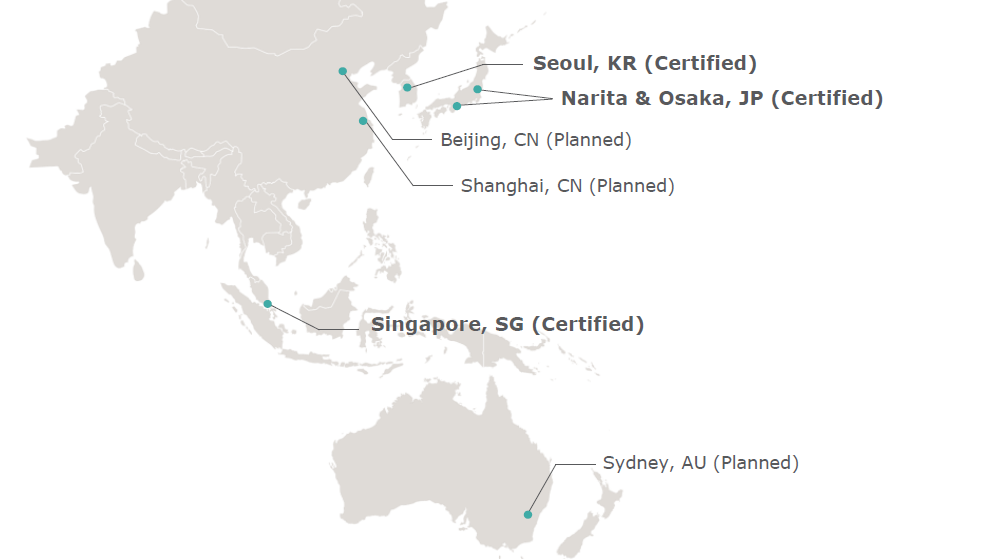
Feb 27, 2026 at 1:25 PMUPS has received three additional IATA CEIV Pharma certifications to strengthen healthcare in Asia. The certifications were awarded for cargo handling at the international airports of Tokyo-Narita (NRT) and Osaka-Kansai (KIX) in Japan, as well as at the international airport of Seoul-Incheon (INC) in South Korea. This recognition confirms UPS’s ability to transport high-quality pharmaceutical products safely and efficiently.
With these new certifications, which complement the award received in 2019 for Changi Airport in Singapore, a total of four UPS cargo handling operations are now internationally recognized. The IATA CEIV Pharma certification ensures that the processes in pharmaceutical air freight comply with global best practices and that UPS Healthcare™ Cold Chain Freight services at these locations meet the highest standards.
Wes Wheeler, President of UPS Healthcare, stated: “The latest certifications provide healthcare decision-makers and our customers in the pharmaceutical and life sciences sectors with the assurance that their products are handled according to the highest regulatory requirements.” This underscores UPS’s commitment to enhancing healthcare in the Asia-Pacific region in line with global quality standards.
Growth in Cold Logistics
Spending on cold logistics is projected to rise to an estimated $21.3 billion by 2024. This growth highlights the increasing demand for specialized healthcare services, particularly in Asia, where established life sciences hubs in Singapore, Japan, and South Korea are thriving. Anita Li, Vice President of UPS Freight Forwarding for North and South Asia, emphasized that UPS has the necessary capabilities to operate a seamless cold chain, which is crucial for protecting the integrity and quality of temperature-sensitive biopharmaceutical products, such as vaccines and clinical trials.
In the future, UPS plans to seek certifications for cargo handling in Shanghai and Beijing, China, as well as in Sydney, Australia.
UPS Healthcare recently introduced a comprehensive range of technologies for the cold supply chain, along with new and expanded global facilities to offer temperature-controlled logistics solutions. By 2025, UPS will have expanded its cold chain storage capacity in accordance with Good Manufacturing Practices (GMP) by approximately 36,000 square meters to support the specialized storage of biologics.
The newly opened brand-GMP depot facilities in Japan and South Korea, along with the ongoing expansion of an existing facility in Sydney, aim to connect the region with global markets for specialized storage, compliance, and handling needs in pharmaceuticals, biotechnology, and medical devices.